iNtRON Biotechnology has gained approval to start an additional Phase 1b clinical trial to test the safety of its antibacterial drug N-Rephasin® SAL200 regarding repeated doses, the company said Friday.
The Korean biotechnology company’s therapy is a novel antibacterial drug for multidrug-resistant (MDR) staphylococcus-associated infections. The global health industry is facing a crisis because of super bacteria resistant to most types of antibiotics, requiring new therapies, iNtRON인트론바이오 said.
According to an Antimicrobial Resistance (AMR) review report, super bacteria are responsible for 30,000 deaths annually in Europe. The AMR warns the number could increase to more than 10 million, costing $100 trillion by 2050.
N-Rephasin® SAL200, also called SAL200, is a first-in-class phage endolysin-based drug proved useful in treating new resistant forms of staphylococci that do not cause bacterial resistance, unlike conventional antibiotics, the company said.
“Although existing antibiotics work through inhibiting, they do not kill bacteria quickly enough, leading to the development of drug resistance. SAL200 has an entirely different mechanism from existing antibiotics by killing germs on contact,” a company official said.
The World Health Organization designated a new name (TONABACASE) for the drug as the first registered phage endolysin in 2015. Endolysin are proteins produced by bacteriophages (“predators” that infect and kill bacteria) that have rapid and potent antibacterial activity.
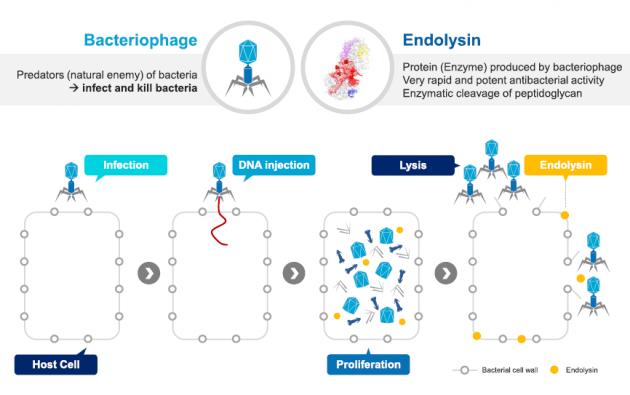
iNtRON Biotech had also gained the Ministry of Food and Drug Safety’s approval to carry out the world’s first endolysin injection study domestically.
Currently, phase 2a clinical trials for SAL200 are underway. The randomized, double-blind, placebo-controlled phase-2 trial held at Seoul National University Hospital and Bundang Seoul National University Hospital targets patients with persistent bacteremia originating from Staphylococcus aureus.
According to the company, the newly approved 1b trial will target 40 healthy male volunteers, administering the drug in incrementally increasing dosages. The trial will have one single-dose group and four repeated-dose groups.
“The phase 1 clinical trial revolved around a single dose. The new phase 1b trial is a multiple ascending dose (MAD) clinical trial, which is a multi-dose treatment,” the company official said. “The global pharmaceutical companies we have partnered with also wanted to confirm the safety of multiple doses, which is why we have decided to go ahead with the 1b trial to further expand the target market for SAL200.”
Industry insiders expect the company will fulfill the unmet demand in the market for antibiotic resistant bacteria and the existing synthetic antibiotic market to solve the problem regarding bacteria’s increasing drug tolerance.
The Methicillin-resistant Staphylococcus aureus (MRSA) “super bug” had a $3.2 billion market in 2013 shifting to more effective drugs, according to IMS Healthcare.
The company is also developing both Gram-positive and Gram-negative bacteria programs to develop a new therapeutic approach to antibiotic resistant bacterial pathogens.
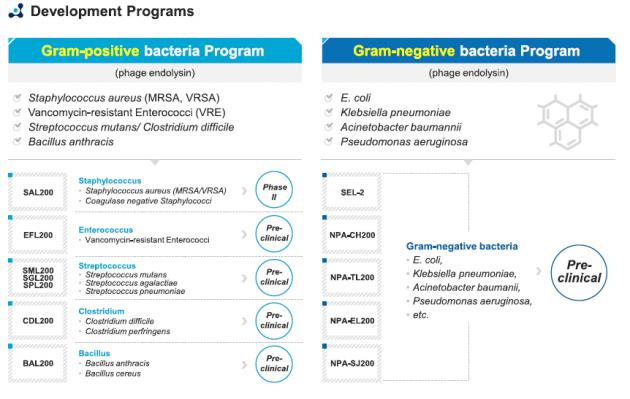
To date, iNtRON has successfully developed five lead BACASEs effective against Gram-negative bacteria such as Salmonella, Escherichia coli, and Klebsiella pneumoniae, the company said.

