Pfizer’s Janus kinase (JAK) inhibitor Xeljanz (ingredient: tofacitinib) lowered the risk of death and respiratory failure in patients hospitalized with Covid-19 pneumonia, a study showed.
The latest results give Pfizer an edge not only as a Covid-19 vaccine developer but a Covid-19 treatment supplier.
On Wednesday, the New England Journal of Medicine published the results of the STOP-COVID study, titled “Tofacitinib in Patients Hospitalized with Covid-19 Pneumonia.”

The research team said severe Covid-19 infection was associated with “an exaggerated immune response driven by interleukin-6, tumor necrosis factor α (TNF-α), and other cytokines in a pattern called ‘cytokine storm.’”
Tofacitinib is an oral JAK inhibitor that selectively inhibits JAK1 and JAK3, with functional selectivity for JAK2. It blocks intracellular transduction pathways after a cytokine is bound to its receptor, and this could indirectly suppress cytokine production, the research team explained.
Tofacitinib can also control the actions of interferons and interleukin-6, reducing the release of cytokines by type 1 and type 17 helper T cells related to the acute respiratory distress syndrome (ARDS).
“Thus, the action of tofacitinib on multiple critical pathways of the inflammatory cascade may ameliorate progressive, inflammation-driven lung injury in hospitalized patients with Covid-19,” the research team said.
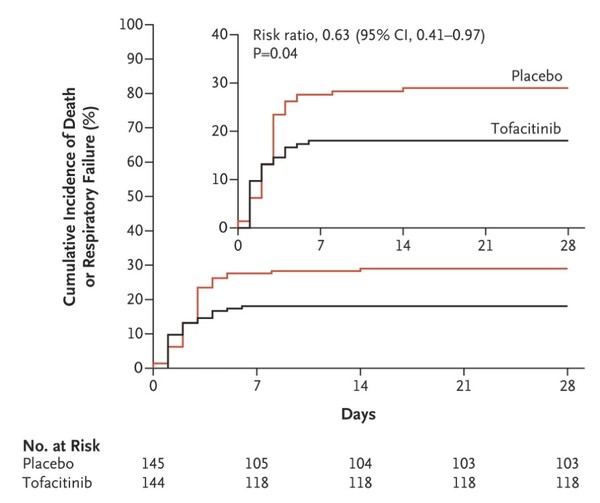
The researchers randomly assigned 289 patients in 15 hospitals in Brazil to the twice-daily Xeljanz 10mg group and the placebo group for up to 14 days or until discharge. The primary outcome was death or respiratory failure during the 28 days of follow-up. The researchers also evaluated death from any cause and safety.
The results showed that the cumulative incidence of death or respiratory failure through day 28 was 18.1 percent in the Xeljanz group and 29 percent in the placebo group. This means that Xeljanz cut the risk of death or respiratory failure by 37 percent.
Death from any cause occurred in 2.8 percent of the Xeljanz group, which was 51 percent lower than 5.5 percent in the placebo group. Serious adverse events occurred in 20 patients (14.1 percent) in the Xeljanz group and 17 patients (12 percent) in the placebo group.
“Among patients hospitalized with Covid-19 pneumonia, tofacitinib led to a lower risk of death or respiratory failure through day 28 than placebo,” the research team concluded. “These effects were consistent regardless of sex, age, duration of symptoms, use of glucocorticoids at baseline, and different levels of supplemental oxygen use at baseline.”
Xeljanz is not the first JAK inhibitor that proved a therapeutic effect against Covid-19.
Lilly’s Olumiant (baricitinib) also shortened the recovery period in patients receiving high-flow oxygen therapy or non-invasive mechanical ventilation in the ACTT-2 trial. Olumiant proved the effect in combination with Gilead’s first antiviral drug remdesivir in the ACTT-1 study.
Based on current evidence, antiviral therapy is likely to be effective in early Covid-19, and hyperinflammatory responses are thought to cause clinical symptoms in later stages of the disease, according to the study. Thus, anti-inflammatory interventions are needed in hospitalized patients with mild, moderate, or severe Covid-19, the research team said.
The results of ACTT-2 and STOP-COVID suggest that “JAK inhibition represents an additional therapeutic option for treating Covid-19 pneumonia in patients who are not yet receiving invasive mechanical ventilation,” the research team added.

