Korea UCB (CEO: Lee Young-joo) said Tuesday the Ministry of Food and Drug Safety has approved its hemophilia A treatment.
Hemophilia is a rare genetic disorder, also called factor VIII (FVIII) deficiency or classic hemophilia, which causes hemorrhages. It is caused by missing or defective factor VIII, a clotting protein. Hemorrhage complications lead to severe edema and pain, arthritis, joint damage, disability, and death.
The World Federation of Hemophilia (WFH) estimates that there are about 400,000 hemophilia patients worldwide.
UCB’s hemophilia A treatment, Eloctate, is a recombinant factor VIII produced by DNA technology. It is the first hemophilia A treatment with a prolonged half-life approved for inhibiting and preventing hemorrhage, pre- and post-operative care, and routine preventive therapy, Korea UCB said.
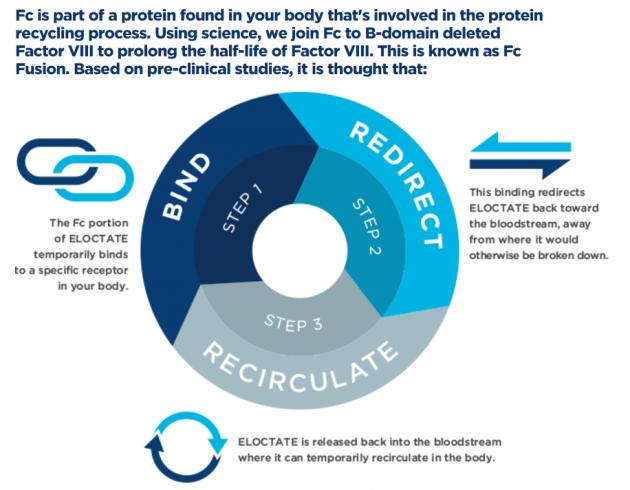
The treatment is administered once every three to five days, a reduction compared to other therapies, according to the enterprise.
The drug was approved based on two phase 3 clinical trials – A-LONG and Kids A-LONG – that proved the drug’s safety and efficacy in 165 hemophilia patients older than 12 years old who received previous treatment, and 71 patients between two to 11 years old, respectively.
The studies showed Eloctate to be useful in the routine preventive treatment and acute bleeding treatment with good safety and tolerability, the company said.
Korea UCB also said its half-life extended gene recombinant hemophilia B treatment, Alprolix (coagulation factor IX, Fc Fusion Protein), gained the ministry’s approval in May as the first factor IX that helps patients prevent bleeding episodes.
The two drugs – jointly developed by Bioverativ, a division of Biogen, and UCB – had gained the U.S. Food and Drug Administration’s marketing approval in 2014. The drugs are currently sold worldwide, including the U.S., Japan, and Europe, the company said.
“We are delighted to be able to offer new treatment options to hemophilia patients through the domestic approval of Eloctate and Alprolix,” said Korea UCB CEO Lee Young-joo. “UCB will continue to improve lives and provide new value in the field of rare diseases such as hemophilia.”

