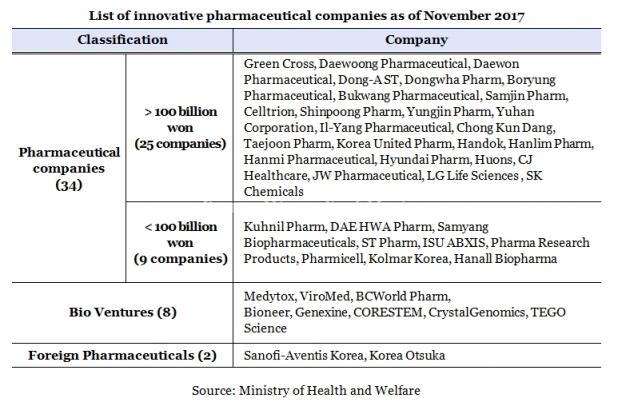
The Ministry of Health and Welfare said it has recertified three pharmaceuticals – Genexine, Huons, and Sanofi-Aventis Korea – as “innovative” as their initial certification is to be expired Wednesday.
The ministry also said that SK Chemicals has moved up a rank in the list of innovative companies.
A committee dedicated to fostering and supporting the growth of the pharmaceutical industry hands out the certification to companies that invest a certain amount into research and development for new drugs, ministry officials said Monday.
The committee is comprised of 15 officials from public and private sectors.
As of this month, there are 34 domestic pharmaceuticals, eight bio ventures, and two multinational pharmaceuticals with the certification, which lasts for three years.
The recertified companies will hold the title until 2020, the ministry said.