Yonsei Medical Center professor tries to help pharmaceutical firms by reducing risks
Businesses get management consulting at great expense. They undergo the inspection of internal state and work out countermeasures according to its results. All this is necessary to prevent massive losses resulting from failure to prepare for risks.
The biggest risk for pharmaceutical companies is the failure concerning the development of new drugs. Statistically, only one in 10,000 potential new medicines proves successful. The success of course leads to enormous financial gains but projects spending astronomical sum often ends up in failure, incurring tremendous losses on developers.
That explains why pharmaceuticals seek counsels from experts in the early stage of developing new drugs. These experts are mostly physicians, knowing well how their colleagues treat patients and how they prescribe medicines in clinical settings. Nonetheless the process of tracking people down and finding related data is not always easy.
So how good would it be for businesses if they could assemble various experts at one place and hear their counsels simultaneously? What if the companies could receive data based on medical treatment information? It would be the icing on the cake for businesses developing new drugs.
And that is what the Yonsei Medical Center is now trying to do. It has recently formed SALTPLUS (Strategic Advisory Leadership Team), a customized consultation group of new drug developing strategy, providing “data-based clinical information and researcher-based counsels.” Its biggest advantage is to let customers make the best use of the medical information, researcher pools and infrastructure facilities accumulated within the institution.
The Korea Biomedical Review met with two key staff members of the team –Professor Shim Jae-yong, who leads SALTPLUS and heads the Global Center of Excellence in Clinical Trials, and Secretary General Kim Dong-kyu, who operates its coordinating center – to ask about their services and plans.
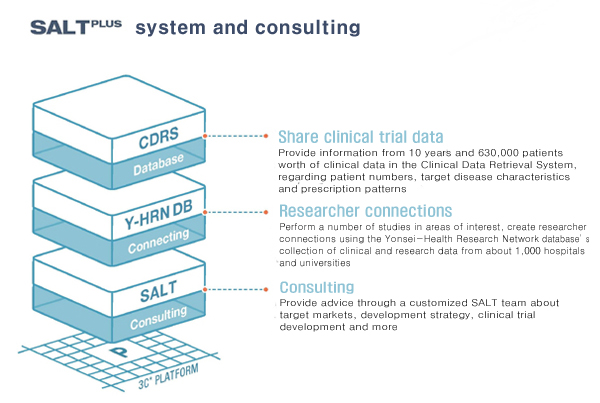
Question: Please explain about SALTPLUS briefly.

Shim: SALTPLUS is a consultation service for developing new drugs based on “real data.” Too many projects end up in failure due to the lack of proper advice at initial stages. This is because medical experts’ insights can be different from corporate viewpoints. Also, physicians who treat patients in the field can have better “marketing sense” than companies do.
SALTPLUS provides clinical information for high-profile diseases based on 6.3 million data sets available at Yonsei Medical Center. We also offer customized consultation that utilizes results from about 1,000 researchers at the university and affiliated hospitals and by forming advisory teams. Therefore, companies can receive consultation from many experts in related fields with real clinical data. SALTPLUS is probably the first consulting team using real clinical data.
Kim: SALTPLUS is the collaboration of the SCI-C consortium, which includes the Severance Hospital, Gangnam Severance Hospital, Inha University Hospital and the Seoul St. Mary’s Hospital of the Catholic University of Korea. SIC-C has provided SALTPLUS consultation, which sets itself apart from others in that it systematically offers counsels based on the hospitals’ clinical data. Each of the four hospitals has experts ready to provide appropriate advice, and their ranges of researchers are very broad. It is our objective to analyze data in depth, provide a wide spectrum of information and go further to win contracts for a clinical experiment.
Q: What was the background behind creating SALTPLUS?
Shim: We created it to promote appropriate consultation for pharmacists who encounter difficulty during the first stages of development, and eventually to reinvigorate the development of new medicines. To put it plainly, they need to take the good first step. The most common mistake of pharmaceuticals stems is their failure to confirm how discriminately their new drugs can supplement (the existing medicines). They have simply visited some physicians to survey how many patients they treat and what medications they use. However, new drug development requires numerous decision-making, from the planning stages and actual development to clinical experiments. It also is an area which requires the investment of lots of money and time. Accordingly, acquiring correct and diverse information is the best way to maximize the effectiveness of research and development.
SALTPLUS possesses about 47 million clinical data sets from 6.3 million patients who have visited the Yonsei Medical Center. We have also created a database from basic medicine and clinical research results attained by about 1,000 researchers at the university hospital. The database consists of about 38,000 theses, 1,700 patents, 20,000 research tasks and 14,000 clinical trials.
Q: To which stage of new drug development can pharmaceuticals receive SALTPLUS’s help?
Shim: We can provide assistance during the primary development process -- from substance selection to initial clinical trials that prove the efficacy of a drug. When pharmaceuticals analyze the target market’s characteristics to select target substance and disease, they can know about the current situation of the market and predict its future through clinical treatment information the university hospital has.
In other words, drug companies can set up a more concrete strategy for clinical development based on the analysis of various information, such as diagnostic criteria of target disease, details of relevant patients (complications and associated diseases), and the kinds of drugs prescribed, provided through the database.
And this is possible for all sorts of new medicines -- first-in-class, best-in-class, and incrementally modified drugs. For instance, when pharmaceuticals develop treatments for cerebral stroke, they first find a candidate substance and develop a new medicine based on it. However, physicians research the stroke itself. That means they have information on what’s going on in the medical scenes. They can confirm what kinds of treatment produce effects and within what time limit to treat a disease, and then provide information on illness and their drugs, new or modified, including their internal resorption and distribution. They can even suggest new research directions.
However, SALTPLUS does not provide pharmaceuticals with the “discovery” phase, not helping them find new substances, in other words. Instead, when the drug companies licensed in candidate substances and develop them, we can make them check at least how many patients are in Korea or whether it is right to introduce the drug of related mechanisms.
Q: Why is consultation necessary in developing new drugs?
Shim: Drug companies hesitate to ask for outside opinions whether to develop candidate substances into treatments, for fear of leaking confidential information even before their official registration. They seek consultants through personal acquaintances and make secret contracts. It is difficult for companies to meet KOL (Key Opinion Leader) and ask their counsels for many reasons. Even if they can, there is no guarantee of success.
But consultation is crucial in developing new drugs. Businesses should seek counsels more systematically and get more responsible consultations. As business corporations invest an enormous amount of money in consultation, pharmaceuticals need to receive experts’ consultation more systematically. However, some makers still seem to have doubts about the need for systematic consultation.
Q: What does the coordinating center do?

Kim: It is a system dedicated to helping the company effectively utilize the range of research resources available in a university hospital. We care most about time because they want to get information within a limited time.
The center can also provide confidential and formalized consultation services. With the Fair Competition Reinforcement Act and the so-called “Kim Young-ran Act,” the laws require documentary evidence for consultation services. In the past, companies met one-on-one with consultants, making integrated management impossible. The center abides by the law in providing consultation service, and ready to submit standardized documents to the government.
Q: How have companies reacted to SALTPLUS? What are they particularly interested in?
Shim: We introduced SALTPLUS toward the end of last year. Both domestic and overseas pharmaceutical companies have expressed interest primarily in our various R&D stages, without too much focus on specific areas like target disease analysis, clinical trial feasibility, the establishment of clinical development strategy and expected number of patients. We are providing clinical medical data to and consulting with researchers, both here and abroad. Target diseases are currently focused on a variety of treatment areas, such as digestive, endocrine, cardiovascular and antibiotics systems.
In the past, it was hard to select researchers for consultation, as we had to go through companies’ sales or marketing teams and find doctors with research achievements in the diseases of our interest. With the use of SALT research database, however, the quality of consultation has improved significantly.
Kim: A company can select singular or multiple consultants from a pre-selected list of optimal consulting candidates. Our customers also show high satisfaction with our system through which they can receive the opinions of various researchers at once. We’ve found that even some non-pharmaceutical companies have expressed interest in grafting SALTPLUS to their business.
Q: It seems like SALTPLUS will serve a guide in developing new drugs.
Shim: SALTPLUS is an idea that had not existed previously. Both consultation and data had existed before, but their combination is an entirely new attempt. University hospitals, in particular, have both researchers and data. We have combined people and information to provide immense value for the development of new drugs. We have also solved the problem of not making the full use of data to protect personal information. The data we provide are for the sake of research and were made unidentifiable. SALTPLUS can provide consultation through all the phases of developing new drugs. If pharmaceutical companies make the right course in the first state, they can reduce the risks of failure. We also expect joint development will be possible.

