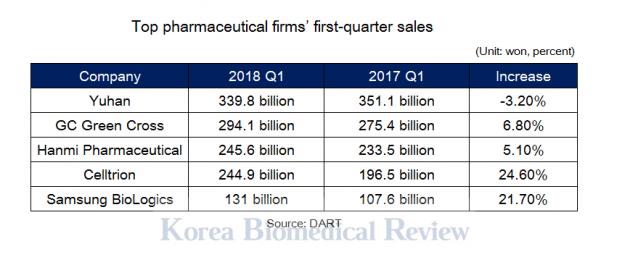
Celltrion recorded sales of 245 billion won ($227 million) in the first quarter of this year, up nearly 25 percent from the same period of last year. Most of the growth came from strong sales of its biosimilars, the firm said.
Celltrion's revenue posting brought it to the front of the biopharma pack regarding revenue, surpassing turnover registered by competitor Samsung BioLogics, which posted 131 billion won in first-quarter sales.
The Korean biopharma firm came behind three leading pharmaceutical firms, such as Yuhan Corp. that posted 339.8 billion won ($314.8 million), GC Green Cross, 294.1 billion won, and Hanmi Pharmaceutical, 245.7 billion won.
Celltrion reported the sharp sales increase was thanks to the continued uptake of Remsima in the European and U.S. markets and expanding market share of Truxima in Europe.
According to the firm, sales of Remsima (known as Inflectra in the U.S.) accounted for a whopping 52 percent of first-quarter sales and grew 224 percent from a year earlier.
Remsima posted 59 billion won ($54.6 million) in sales in the first three months of 2018, with about 17.4 billion won sold in Korea, according to IQVIA data.
Truximas sales contributed 23 percent to the total revenue.
“We are actively proceeding with European market entry with three fast-mover products, beginning with the European sales of Herzuma in early May. We will work in earnest to make Truxima a first mover in the U.S. market,” a Celltrion official said.
Celltrion has been one of the leading biosimilar firms in Korea having gained the world’s first approval for Remsima - Janssen Biotech’s Remicade biosimilar – in Europe and the U.S. The company is developing a subcutaneous injection of Remsima and plans to launch Herzuma in Europe soon.

