Multinational pharmaceutical firms had more phase 3 trials underway in Korea while domestic drugmakers had more phase 1 trials in the past five years, data from the Ministry of Food and Safety showed.
Clinical trials, comprised of mainly four stages, are essential to explore whether a treatment is safe and effective for people. All therapies must go through the four clinical trial stages to be approved by the health regulators.
In 2017, global pharmaceutical firms had 178 P3 trials underway in the country while domestic pharmaceutical firms had 31. The number of phase 3 trials for global pharmaceuticals rose 30.9 percent from 136 studies in 2016 to 178 tests last year.
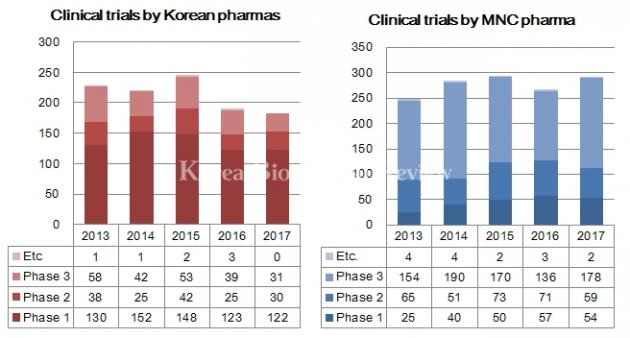
As for phase 1 studies, Korean pharma had 122 trials while multinational corporations had 54 last year.
Phase 1 trials are the first drug safety screening tests done on humans. The primary objective of the small study (20 to 80 people) is to determine the safe dosage range.
Phase 3 trials are large-scale scales that range from a minimum of 100 to 10,000 people that aim to confirm efficacy, evaluate effectiveness, and monitor side effects of therapy while also possibly comparing it to other treatments.
“More domestic pharmaceutical companies seem to be focusing on developing new drugs. The number of phase 1 and 2 trials is also increasing because Korea has been raising its competency regarding clinical trials,” said an official at Korea Research- based Pharma Industry Association (KRPIA).
“Globally, new drug development projects have decreased in number. Despite the drop, Korea has maintained its level relatively well in the number of clinical trials,” the official added.
According to the Korea National Enterprise for Clinical Trials, the total number of clinical trials rose from 628 in 2016 to 658 trials in 2017, indicating a 4.8 percent increase.

