Naxozole by Hanmi Pharm has kept No.1 since 2013
‘Vimovo (ingredient name: Naproxen+Esomeprazole magnesium trihydrate) made by AstraZeneca to treat osteoarthritis is struggling in the Korean market. The drug lost No.1 place in sale to incrementally modified drug, ‘Naproxen+Esomeprazole strontium tetrahydrate‘(Korean name: Naxozole) made by Hanmi Pharm for 3 consecutive years in Korea.
Vimovo is the drug to combine Naproxen used in NSAIDs (nonsteroidal anti-inflammatory drugs) to PPI (Proton Pump Inhibitor) Esomeprazole (product name: Nexium) to treat gastric ulcer. Side effects of NSAIDs such as heartburn can be reduced.
Since the release of Vimovo in 2012, market of 'NSAIDs+PPI' has been growing every year. In Korea, besides Naxozole, 6 generics such as ▲Naxen S Tab by Chong Kun Dang pharmaceutical, ▲ Esoroxen by Korean Drug Co.,Ltd., ▲ Synflex Safe Tab by PMG Pharm, ▲ Naprazole by Alvogen Korea , and ▲ Nafmed by SK Chemical have been released.
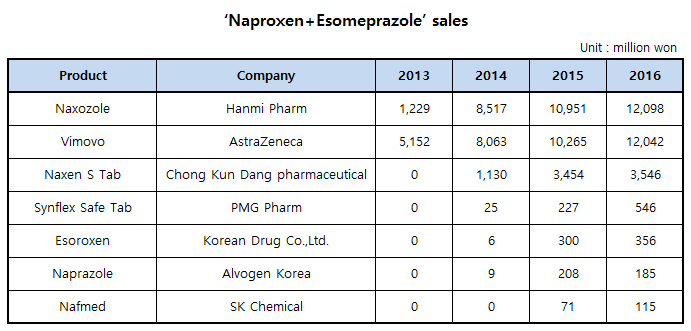
According to the statistics service of medical supplies UBIST, the market size of the 'NSAIDs+PPI’ recorded about 28.8billion won in 2016. In the market, Vimovo and Naxozole play leading roles in sales. The sales of Vimovo and Naxozole stood at about 12billion 98million won and about 12billion 42million won respectively in 2016.
Even though the original drug Vimovo was released ahead of Naxozole in the Korean market, it always lost No.1 place to Naxozole. The sale of Vimovo in 2013 was in the first place (5.1billion 52million won), but has lost its place to Hanmi Pharm since 2014.
But there is possibility of Vimovo to retake the place because the increase rate in sale is higher than the rate of Naxozole. The sale of Naxozole and Vimovo in 2016 increased 10.5% and 17.3% from a year earlier respectively.

