Korean botulinum toxin maker Medytox said the Chinese regulator ordered the review suspension on its product Meditoxin only once, not multiple times.
The clarification came after Korea Biomedical Review reported on Monday that the Chinese drug authorities were reviewing additional data and that the regulator slapped multiple review suspensions on Meditoxin. (Related news: Medytox’ botulinum toxin may face approval delay in China)
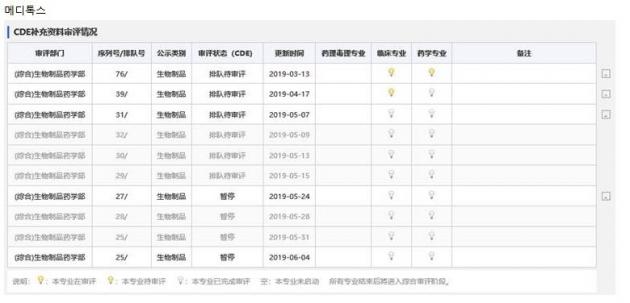
The company said the multiple displays of the review suspension on Meditoxin on Yaozh.com, a Chinese online site for searching drug approval progress, were a result of an update of the website.
“The online site updates drug review progresses every week, and the content can change following status change or a change of a month,” an official at Medytox said. “We didn’t get review suspension twice, but the display appeared twice as the month changed from May to June. The suspension as of May 24 remains unchanged.”
Elaborating its position further, the company disclosed the Chinese site’s status window on the Medytox product.
“It is true that the Chinese regulator requested us to submit additional data one tie. However, we submitted all the necessary data on March 13, and the review was in progress,” the official said. “As the assessment on additional data has been completed on May 24, the window showed ‘suspension’ for a next step to be decided.”
Suspicions that the company received multiple review suspensions were not true and that the evaluation was at the final stage, Medytox said.
“The site predicts we will receive approval on July 27. We are at the final stage of the review. We do not have any issue in the approval process now,” the Medytox official said.

