GC MS said that it has registered a patent for a technology that can detect detecting influenza virus in the United States. This is the first time that GC MS has obtained a patent in the U.S.
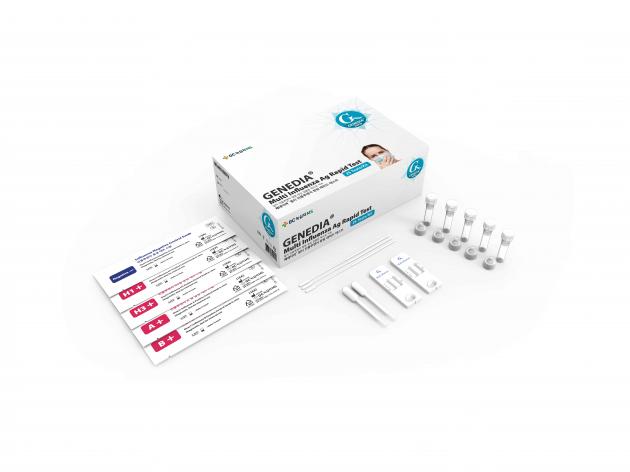
The patent is a technology for detecting influenza viruses through a multi-influenza diagnostic kit. By collecting sputum or throat secretions, the kit can detect various influenza viruses, such as type A, B, H1N1 (swine flu), and H3N2.
The company explained that it had proved its technological competitiveness in the multi-influenza diagnosis sector by acquiring the U.S. patent after it obtained the same patent in Korea.
"The patent acquisition is significant as it is the first patent that we have registered in the U.S.," GC MS CEO Ahn Eun-uk said. "We will continue to increase our product competitiveness through continuous R&D investment."
GC MS has been marketing the "GENEDIA multi influenza Ag rapid test" that uses the patented technology in Korea since 2014. The company is also exporting the product to Mexico and Malaysia.

