“I heard there is a quick test for meningitis. Where can I get it?”
A patient receiving meningitis treatment recently called Korea Biomedical Review (KBR). He said he learned about the existence of a quick test after reading an article of The Korean Doctors’ Weekly, the sister paper of KBR, that the test, which could diagnose meningitis and the cause within an hour, could be unavailable because of the low reimbursement.
The patient said that even though he was getting treatment, he did not know whether his disease was bacterial or viral.
Meningitis treatment differs depending on the cause. It usually takes about a week to confirm the cause. Bacterial meningitis could cause death in a day or two due to a sudden worsening of symptoms. Doctors can use antibiotics before determining the cause.
KBR could not recommend any hospital for the patient because hospitals have stopped running the one-hour test for meningitis since the test received the health insurance coverage.
The “FilmArray” test is a polymerase chain reaction (PCR) system that shortened the diagnosis time for meningitis and encephalitis from over one week to about an hour. Since the government granted the health insurance benefit for the FilmArray test in September 2018, however, clinicians have seldom run the test. It was because the reimbursement for the test was lower than the cost of the test.
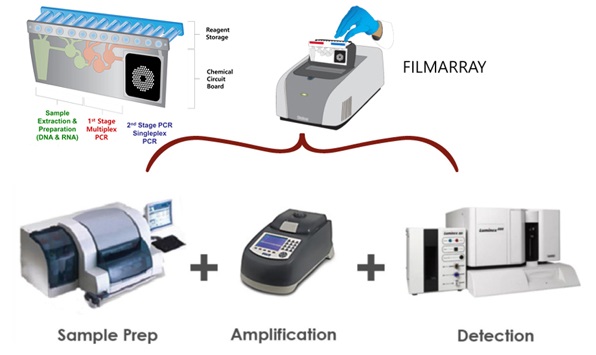
A hospital receives 93,000 won ($78.3) for a FilmArray test from the government, while bioMérieux Korea imports the test panel (kit) from its French headquarters for 100,000 won and supplies it for the hospital for 180,000 won. BioMérieux Korea said it could not lower the price because it lends and sells the test panel through a contractor. The company did not lower the price, and the hospitals were forced to discontinue the tests because they made losses if they provided the service.
According to bioMérieux Korea, the sale of FilmArray panels started to plunge immediately after the reimbursement. For a year from December last year, the company has sold no product. This means that there is no local hospital that can confirm the cause of meningitis and encephalitis within an hour.
‘Decision-mediating review too slow’
As the issue of the discontinuation of the FilmArray test became public, the health authorities said they would reconsider the reimbursement rate. However, the review is progressing too slowly, experts said.
The medical community has requested a review on the decision of the reimbursement for the FilmArray test, but the review is still ongoing for nine months.
The Korean Society of Pediatric Infectious Diseases, the Korean Society of Neonatology, and the Korean Child Neurology Society submitted a request for mediation of the decision to the Health Insurance Review and Assessment Service (HIRA) on Feb. 28 and sent a letter requesting early deliberations in August and September.
Hospitals that once operated the FilmArray test also sent a letter to the Ministry of Health and Welfare to resolve the issue of the reimbursement quickly so that patients could receive the fast test for meningitis.
“There is no better test than a FilmArray test. Two hours will be enough to identify the cause of meningitis,” said Nam Sang-wook, vice president of the Korean Child Neurology Society and a pediatrics professor at the Pusan National University Yangsan Hospital. The test does not bother the patient at all, and knowing the causative organism could help physicians start treatment accurately and quickly, he added.
Nam noted that if it is unclear whether meningitis is bacterial or viral, the patient has no other option but to receive antibiotics. Otherwise, a side effect could develop, or the patient could even die, he said.
Before the FilmArray became reimbursable, the Pusan National University Yangsan Hospital used to run the test for about 100 patients a year, according to Nam. At that time, patients could choose whether to use the test or not. However, after the reimbursement, the supply of the test panels stopped, and the hospital could not use the test at all, he said.
“We requested the decision mediation review, but the progress was too slow. We even submitted a petition to the health and welfare ministry,” Nam said. “For patients, we should be able to use the FilmArray test as soon as possible. It is not a matter of choice.”
HIRA says it is still considering
As HIRA is taking a cautious stance on the issue, it would be hard to see the review result within this year, observers said.
An official at HIRA said the authorities were discussing the matter with data that could be monitored. “We are trying to conclude in a desirable direction,” he said.
As the process of the FilmArray test panel supply is complicated, there is a gap between the supply price and the actual price, the official said. Considering the price gap, HIRA is trying to adjust the reimbursement, he added.
The official also said that he could not predict when HIRA will announce the review result, as the agency was still discussing the matter.

