AstraZeneca said Lynparza (ingredient: olaparib) has become the first poly ADP ribose polymerase (PARP) inhibitor to improve the overall survival of ovarian cancer patients in a phase-3 trial.
The company released the final results of the SOLO-2 study, which compared the Lynparza maintenance monotherapy with placebo in patients with platinum-sensitive relapsed ovarian cancer with BRCA 1/2 mutations, during the virtual scientific program of the 2020 American Society of Clinical Oncology (ASCO) Annual Meeting, on Wednesday.
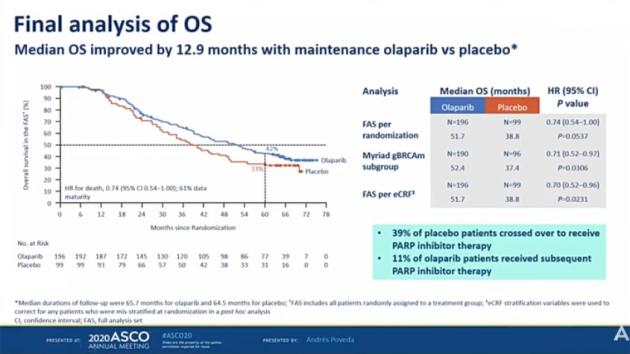
According to data unveiled for the press first, Lynparza extended OS by 12.9 months and reduced the risk of death by 26 percent, compared to placebo, during a five-year follow-up.
“This study confirms that the PARP inhibitor olaparib should be the standard maintenance therapy for patients with BRCA-related relapsed ovarian cancer responding to platinum-based chemotherapy,” said ASCO Chief Medical Officer and Executive Vice President Richard L. Schilsky. “(It is) a significant advance for women with cancer that has a historically poor prognosis.”
Participants of the study previously received at least two lines of chemotherapy and had cancer that was responding to recent platinum-based chemotherapy. The researchers randomized them into two groups – 196 in the Lynparza group and 99 in the placebo group.
At five years of the follow-up, 42.1 percent of Lynparza-treated patients were alive, versus 33.2 percent on placebo. At the median follow-up of 65 months, 28.3 percent of patients who received Lynparza were alive, compared with 12.8 percent of those with the placebo.
The study allowed the cross administration, and 38.4 percent of the placebo group crossed over to Lynparza treatment.
“A median overall survival improvement of nearly 13 months is impressive in ovarian cancer and brings a substantial benefit to our patients,” said lead author Andres Poveda of Initia Oncology, Hospital Quironsalud, in Valencia, Spain.
With the addition of overall survival data, this study helps to usher in a new era of personalized medicine for women with this difficult-to-treat cancer, he added.

