AstraZeneca’s Tagrisso (ingredient: osimertinib), targeted therapy for lung cancer, has won recognition as the first-line standard treatment for epidermal growth factor receptor (EGFR)-mutated lung cancer, after proving that it can extend survival, compared to conventional tyrosine kinase inhibitor (TKIs).
Lung cancer experts worldwide discussed how to overcome the limitation of Tagrisso as first-line therapy in lung cancer at an online oncology meeting.
Professor Ahn Myung-ju at the Department of Hematology and Oncology of the Samsung Medical Center chaired the colloquium session of the European Society for Medical Oncology (ESMO) Virtual Congress 2020 on Wednesday. The participants discussed the present and future of the EGFR-mutated non-small cell lung cancer (NSCLC) treatment.
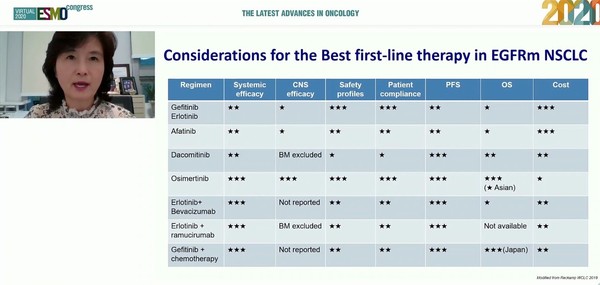
Ahn opened the session with her presentation on “Is Osimertinib the Best Single Agent If 1st Line.”
Given progression-free survival (PFS), overall survival (OS), central nervous system (CNS) activity, safety, and patient compliance, physicians prefer osimertinib as the first-line therapy, Ahn said. However, osimertinib’s acquired resistance is inevitable, she said. Considering its heterogeneity, physicians need to use an optimal sequential treatment strategy or combination treatment to improve OS, she emphasized.
At the meeting, health experts discussed two options – using a TKI with chemotherapy and the combination of a TKI with an anti-angiogenic agent.
Dr. Ross Soo at the National University Cancer Institute in Singapore said Iressa (gefitinib), the first-generation TKI, combined with chemotherapy, showed the same potency as Tagrisso alone in the first-line treatment.
Soo cited study results to say that the PFS in the combo treatment group was 16-21 months, similar to 18.9 months of the Tagrisson alone group. The OS of patients treated with the combo was 50.9 months, much longer than 38.6 months of those treated with Tagrisso alone, he said.
Soo pointed out that Tagrisso had low cost-effectiveness. The treatment costs about $16,000 per month per patient, but studies in the U.S., Brazil, and China have shown that Tagrisso as the first-line therapy was not cost-effective, he said.
“Gefitinib, in combination with chemotherapy, is available and accessible easily. Osimertinib is another option in patients with EGFR T790 mutation, but it offers poor medication convenience due to frequent hospital visits and intravenous administration, and serious side effects,” Soo said.
Professor Martin Reck at the Grosshansdorf Lung Clinic in Germany, who co-chaired the discussion, said the combination of a TKI and an anti-angiogenic agent has the potential to become a new standard of care.
Reck explained that EGFR and VEGFR receptors have a common mechanism and that the combination of two drugs that inhibit them can create synergistic effects. “Preclinical test results showed that VEGF inhibitors could delay resistance associated with T790 mutation,” he said.
Reck backed his claim with another study results that tested first-generation drug Tarceva (erlotinib) combined with VEGF inhibitor Avastin (bevacizumab) versus erlotinib alone.
The phase-3 BeTa Lung study, which compared the combo of erlotinib and bevacizumab group with erlotinib alone in second-line treatment of NSCLC patients, showed that the OS hazard ratio was 0.44 in the sub-group analysis of EGFR-mutated patients, he said.
In her closing comments, Professor Ahn said physicians could use various options for the first-line treatment of EGFR-mutated NSCLC. Not only first- to third-generation EGFR-targeting TKIs but the combo of erlotinib with bevacizumab or ramucirumab, or the combo of gefitinib and chemo, is available, she said.
The U.S. National Comprehensive Cancer Network (NCCN) also recommend these therapies, except for gefitinib plus chemo, as a preferable treatment for EGRF-mutated NSCLC patients, Ahn added.
When choosing the first-line treatment, physicians should consider each drug’s strength and weakness, in addition to systemic effect, CNS effect, PFS, OS, safety, and patient compliance, she said.

