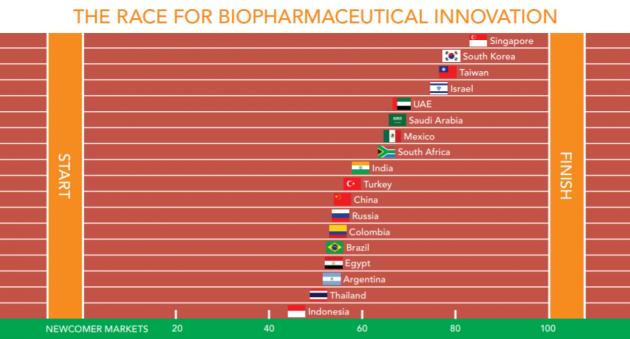
The Korean biopharmaceutical industry ranked second only to Singapore regarding competitiveness and innovation among emerging countries, according to the U.S. consulting group Pugatch Consilium.
Korea has a relatively small biopharmaceutical market, estimated to be around 2 percent of the global market with a value of $17 billion, and a 0.1 percent annual growth rate, according to data from the Korea Drug Development Fund (KDDF).
“Although the market is growing slowly, both the government and public sector are looking at the industry as the future growth engine,” said KDDF Chief Science Officer Kim Sung-chun while presenting at the Global Alliance Project with Novo Nordisk conference Monday.
“The reason for [biopharmaceutical] market stagnancy is that most Korean biopharmaceutical companies are overwhelmed by generics and incrementally modified drugs. Prices are also controlled by the government,” the official added.
To promote growth in the sector, three ministries – the Ministry of Science and ICT, Ministry of Trade, Industry and Energy, and the Ministry of Health and Welfare – have continually expanded research and development to increase national competitiveness, the official said.
The government invested $206.5 million into biopharmaceutical research and development in 2016, while establishing two main programs to foster new drug development.
Notably, the Ministry of Health and Welfare established the Korea National Enterprise for Clinical Trial (KoNECT) in 2007, which is a network hub among clinical hospitals and pharmaceutical companies.
It subsequently created the Korea Clinical Trials Global Initiative (KCGI) in 2016 as a collection of global clinical trial centers operating in five large Korean hospitals, including Seoul National University Hospital, Yonsei Severance Hospital, Asan Hospital, Inje University Busan Paik hospital and Samsung Medical Center.
And the efforts brought fruition, according to KDDF data. Data showed the number of clinical trials in Korea increased 20 times since the turn of the century, with around half of them conducted by local pharmaceutical companies.
Out of the total 675 clinical trials in the country, local pharmaceutical companies carried out 379 of them while multinational pharmaceuticals conducted 296 tests, the official said.
“There is a well-established clinical infrastructure environment in Korea. The country has very big cities and hospitals, which means it is easy to include patients in clinical trials. Also, we have excellent personnel contributing to a great environment,” he added.
The Korea Drug Development Fund is a government-funded organization that supports new drug development across value chains from discovery to clinical trials, the official said. The agency has entered into licensing agreements for 21 pipelines worth $3.5 billion as of July 2016 and funding 116 research projects worth $130 million.

