The Korean Diabetes Association unveiled its latest guidelines for diabetes therapy, citing newest clinical trials and documents, on Thursday.
KDA made public its new guidelines on treating type-2 diabetes at a news conference on the sidelines of the 2017 International Congress of Diabetes and Metabolism at Grand Hilton Seoul Hotel, Seoul.
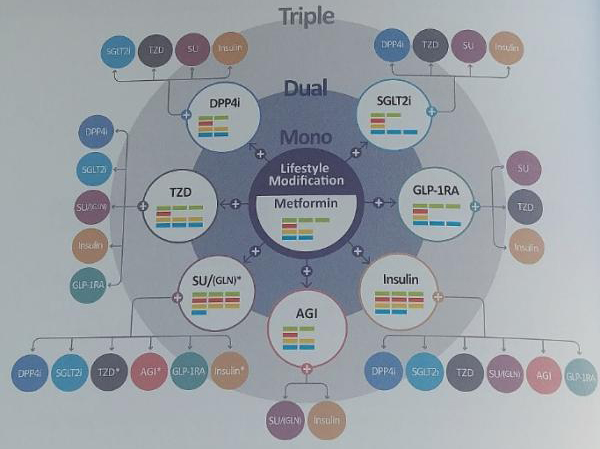
It divided the guidelines into two types – one for orally administered drugs and the other for insulin-targeted therapy. In the case of oral medication, KDA recommended first considering a drug’s efficacy on lowering blood sugar, the risk of hypoglycemia, weight changes, chance of developing cardiovascular diseases and evidence of efficacy through clinical tests.
The guidelines set four recommendations on diabetes treatment; “A” for those proved by a randomized controlled trial (RCT) or having clear evidence through metabolism analysis; “B” for those with credible evidence with a well-carried-out cohort study or a comparative patient-control group research; “C” for those with possible evidence from an unreliable institution’s randomized clinical trial or with some evidence from an observation study and case report; and “E” for those backed by KDA experts’ recommendation without evidence.
KDA categorized the guidelines by affiliation, not by individual drugs. For a mono oral medication, KDA cited metformin as recommendation “A” for early treatment. If metformin is not applicable, other appropriate medication can be selected among those under the recommendation “E.” If the patient’s blood sugar is not controlled by a monotherapy, another drug with different action mechanism under the recommendation “A” can be chosen. Some patients can receive dual therapy under the recommendation “B” after the diagnosis.
A monotherapy can be boosted to the maximum level, but KDA recommends early adoption of dual therapy due to side effects such as failure to control blood sugar. In the case of dual therapy, KDA recommended also considering the drug’s efficacy on lowering blood sugar, risk of hypoglycemia, weight changes, and chances of developing cardiovascular diseases.
In a triple therapy, the drug’s action mechanism, interaction with other drugs, costs, and compliance should be considered, the KDA said.
If a dual treatment fails, KDA recommends insulin therapy, but the treatment can also add or change to a drug in another affiliation.
Putting metformin in recommendation “A” for early treatment was attributed to the drug’s proven safety over the extended period and its cost benefits, KDA said.
The American Diabetes Association (ADA) and the American Association of Clinical Endocrinologists (AACE) also prefer metformin as the first-line therapy for diabetes, but AACE says other drugs can also be used.
“KDA recommends metformin as the first-line treatment, but other therapy can be used in case of digestive disorders or kidney dysfunction,” said Choi Kyung-mook, director of the KDA’s Committee of Clinical Practice Guideline. “Many guidelines overseas also recommend metformin as the first-line treatment, if patients do not suffer any side effect.”
Choi also said that if the use of metformin is impossible, all second-line therapy can be used as first-line. “Indicating metformin as the first-line therapy does not always mean patients should use metformin no matter what,” he said.
The KDA guidelines also updated the early treatment principle, emphasizing the importance of the patients’ efforts to change their lifestyles along with drug treatment.
In the early treatment, they should consider the drug’s efficacy, side effects, costs and the patient’s clinical features, the KDA said, adding that drug therapy can start immediately after the diagnosis and the patient should simultaneously make an effort to change lifestyle, it said.
The latest guidelines mostly reflected local studies because efficacies of diabetes drugs can differ depending on ethnic groups. However, the KDA said the guidelines should be upgraded with more details based on more local researches because there are not enough local studies yet.
“The guidelines were designed in a way that doctors can decide which drug should be used for which step,” Choi said. “We placed it from mono to dual to triple therapy. Although we didn’t put individual drugs in order, we evaluated them in affiliated groups with bar graphs to indicate efficacy on lowering blood sugar, the risk of hypoglycemia, weight changes and development of cardiovascular diseases.”

