Researchers at Severance Hospital have discovered a targeted anticancer therapy candidate for a type of refractory stomach cancer called epithelial-mesenchymal transition (EMT)-subtype gastric cancer.
The findings are expected to drive forward therapy development for other refractory cancers the researchers said.
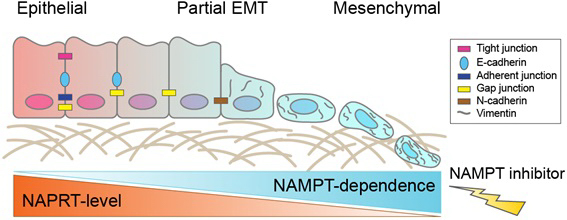
EMT in gastric cancer is the process of epithelial cancer cells turning into tumor cells that have mesenchymal cell characteristics. This transformation makes the cancer cells resistant to anticancer therapies and leads to metastasis.
Among stomach cancer patients, those with an EMT-subtype of gastric cancer had a worse prognosis with their five-year survival rate standing at lower than 30 percent. Those with an EMT-subtype of gastric cancer accounted for 15 to 43 percent of all stomach cancer patients, according to the Asian Cancer Research Group.

The research team led by Professor Kim Hyun-seok from Yonsei University College of Medicine’s Severance Biomedical Science Institute and the team led by Professor Cheong Jae-ho from Yonsei University Hospital’s Surgery conducted research to find a solution.
One of the significant barriers to treating refractory or metastatic stomach cancer is the EMT phenomenon for which no effective therapy exists as of yet, according to Yonsei.
The researchers used a self-developed biomarker and anticancer drug development platform to screen 1,345 compounds for their ability to kill cells with the EMT signature.
Researchers found that FK866, which inhibits nicotinamide phosphoribosyltransferase (NAMPT) function, selectively killed cancer cells in EMT subtypes of gastric cancer where NAPRT enzymes were down-regulated.
NamPT and NAPRT are complementary enzymes involved in the biosynthesis of Nicotinamide adenine dinucleotide (NADs), which is essential for creating energy required for cell survival. Therefore, inhibiting NAMPT function blocked cancer cells of EMT subtype from generating energy, thereby killing them selectively.
The efficacy of FK866 was also confirmed in animal experiments, the researchers said. The team used an independently developed, patient-derived stomach cancer transplant animal model called PDX to demonstrate that FK866 inhibits tumor growth in EMT-subtypes of gastric cancer.
In addition to the anticancer drug candidate for EMT-subtype gastric cancer, the researchers said they developed an accompanying diagnostic method.
Immunochemical staining analysis of 942 patients at Severance Hospital confirmed that the disappearance of E-cadherin protein, which inhibits cancer metastasis, is highly correlated with NAPRT protein deficiency. Therefore, patients with tumors with low NAPRT intensity according to immunohistochemistry will be eligible for the therapy.
Big data analysis also showed the down-regulation of NAPRT expression was found not only in gastric cancer but also in EMT subtypes of the colon and pancreatic cancer. Researchers said that the NamPT inhibitor could be applied to a variety of intractable cancers.
“The research is significant for suggesting a targeted therapy and accompanying diagnostic method not only for stomach cancer but also colon and pancreatic cancer,” Professor Kim said. "We are working on a follow-up study to develop a new drug candidate that maximizes the anticancer effect of FK866 and minimizes adverse effects.”
The findings from the study supported by the Ministry of Health and Welfare’s research-centered hospital fostering R&D project were published in the online edition of Gastroenterology.

