The Ministry of Food and Drug Safety said it has approved two additional artificial intelligence based medical devices – Lunit’s Lunit Insight and JLK Inspection’s JB-01K – on Aug. 14.
The approval comes after the ministry approved the first AI-based medical device, VUNOmed-BoneAge, in May.
The licensed products went through the ministry’s new guidelines for approval and examination of medical devices that use big data and artificial intelligence technology. Both devices received customized support and received approval within 60 days of submitting their applications.
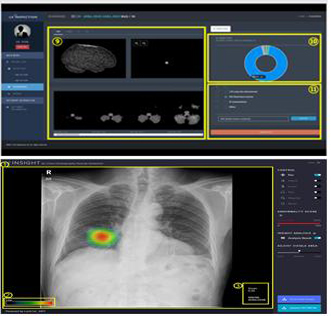
The Lunit Insight inputs and analyzes the chest images of a patient’s X-ray and displays the degree of the suspected pulmonary nodule, which in turn helps doctors diagnose pulmonary nodule.
In the past, the diagnosis of pulmonary nodules needed CT images. With the new technology, however, the company expects that it will reduce costs for CT and other imaging.
According to the ministry, the device showed 94.3 percent accuracy in pulmonary nodules compared to the 89.5 percent when using conventional methods.
JLK’s JBS-01K helps doctors to make an appropriate treatment option for patients with cerebral infarction by providing them with cerebral infarct pattern using the patient’s magnetic resonance imaging (MRI).
During clinical trials, the device showed similar accuracy (58.3 percent) to diagnoses made by doctors (54.4 percent).
“We will do our best to create a customized regulatory environment to enable the development of advanced medical devices that use artificial intelligence, virtual reality (VR), and augmented reality (AR),” a ministry official said.

