The government ordered Korea Abbott to recall its hepatitis B antibody reagent amid concerns that some of the products could show inaccurate test results.
The reagent tests whether a person had been infected by current or past hepatitis B virus by checking the antibody to he patitis B in the serum.
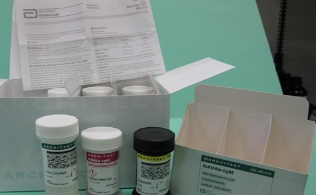
The Ministry of Food and Drug Safety said it ordered Abbott to recall automated immunoassay analyzer Architect HAVAb-IgG, as some of the products did not yield accurate testing results.
A total of 160 products are on recall, with manufacturing numbers “90356LI00,” “90228LI00,” and ”90228LI00.”
Testers must immediately stop using the reagent with the cited manufacturing numbers and dispose of all inventories according to laboratory procedures. With the manager of a test room or a medical supervisor, testers should also review the recall guide offered by Abbott Korea and check whether they need to review the patients’ test results.
Abbott Korea said company officials would visit test rooms, notify the recall, and recollect the products.

