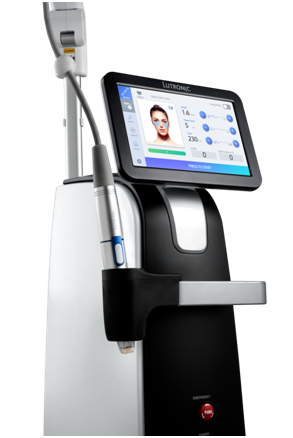
Lutronic, a leading Korean aesthetic company, said Thursday that the U.S. Food and Drug Administration has approved its latest aesthetic laser, Lutronic Genius.
Lutronic Genius is the first product made in the U.S. with features that make it easy to conduct personalized procedures and has an Intelligent Care System that automatically adjusts the optimized energy level.
The system follows the company’s product philosophy – innovative, intuitive, dependable and effective, Lutronic said.
“The new machine expects to receive industry attention regarding both functional and design aspects,” a company official said. “The machine is manufactured based on Intelligent Care System gives more stable energy control through delicate real-time fine adjustment sensor and feedback function.”
Lutronic U.S. COO Lee Pannell also said, “As Lutronic Genius is the first product developed through collaboration between Lutronic Korea and U.S. researchers, the company expects that it will provide superior results for both practitioner and patients.”

