Mediheroes, a medical platform startup, said Tuesday that among the 275 new coronavirus diagnostic kits, Korean products are used the second-most in the world after Chinese ones.
According to the Foundation for Innovative New Diagnostics (FIND) website, one of the World Health Organization's partners for assessment of diagnostic technology, 275 COVID-19 diagnostic kits are in use worldwide. Several dozens of products are being developed as well.
Among the kits, 143 molecular diagnostic kits of 131 organizations are in commercial use now. China manufactures 57 of them, followed by Korea’s 15, and the U.S.’s 14.
As of Tuesday, 22 COVID-19 diagnostic kits from as many manufacturers had won the emergency use authorization (EUA) from the U.S. Food and Drug Administration (FDA). However, none of the Korean products were on the list. According to local media reports, there were controversies on March 28 over the Foreign Ministry’s use of the term “provisional” or “preliminary” approval from the FDA.
According to the FDA’s “Policy for Diagnostic Tests for Coronavirus Disease-2019 during the Public Health Emergency” on its website on March 16, the U.S. regulator allows the use of test kits that are supplied by not only the manufacturers of COVID-19 testing kits but also those purchased by laboratories certified under the Clinical Laboratory Improvement Amendment, and those approved as research use only (ROU).
That means made-in-Korea diagnostic kits that have not won FDA’s emergency use authorization are likely to be used in the U.S. now, according to Mediheroes. Besides, testing centers capable of conducting highly difficult diagnoses can directly purchase diagnostic kits for research use and can screen suspected patients.
Mediheroes said 15 days are enough to get emergency use approval from the FDA. Therefore, other Korean diagnostic kits are expected to receive emergency use approval from the FDA, aside from the three test kits that the FDA had already approved.
According to the Memorandum on Expanding State-Approved Diagnostic Test released announced by President Donald Trump on March 13, the U.S. had authorized each state under the responsibility of its own to develop test kits and use them to test patients regardless of FDA's approval. Moreover, each state is encouraged -- not forced -- to share COVID-19 test results with the FDA.
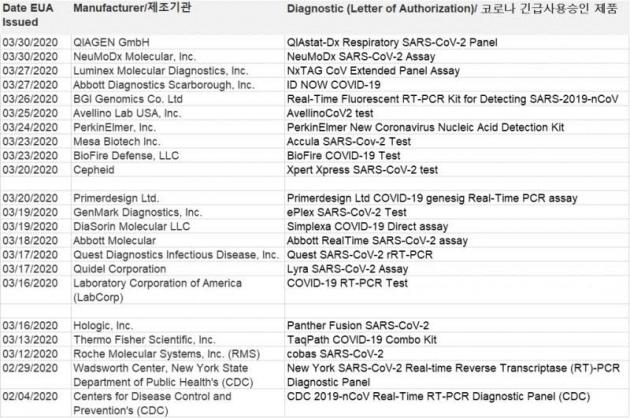
In the Emergency Use Listing updated by the WHO on March 20, one can confirm, aside from the 22 U.S. test kits with an emergency use approval, those made in Korea, Brazil, Singapore, and Australia are being used.
Five Korean test kit manufacturers -- Seegene, Solgent, Kogene Biotech, SD Biosensor, and Biosewoom –- approved by the Ministry of Food and Drug Safety are listed on WHO's COVID-19 Listing in IMDRF (International Medical Device Regulators Forum) jurisdictions.
However, the FDA states that the COVID-19 diagnostic products in use in the U.S. and other countries are not officially approved but won the nod only for emergency use. Those products will not be able to use after the emergent situation ends.
“Korean makers should not remain complacent with a temporary boom of their products, but use the ongoing crisis as an occasion to establish global supply networks for their products,” the platform said.

