Novo Nordisk’s Saxenda is facing fierce competition to keep its dominance in the local obesity market after Alvogen Korea launched Qsymia developed by Vivus, industry sources said Friday.
Novo Nordisk had had the lion’s share in the domestic market until recently. The company sold 42.5 billion won ($35 million) worth of Saxenda last year, accounting for 32 percent of the 134.1 billion won market. The drug recorded an explosive sales growth rate of 466 percent from 2018, which stood at 7.5 billion won.
Since Qsymia of Vivus came to the market, however, Novo Nordisk has been quickly losing its market share as the new entrant emerged as the runner-up in just months.
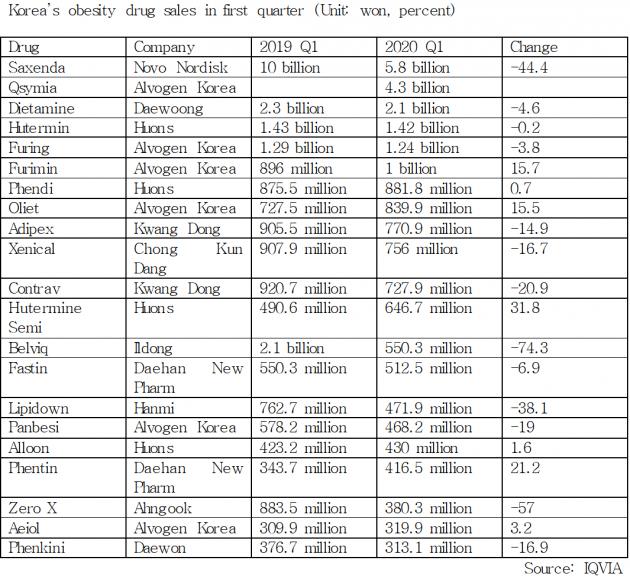
According to IQVIA, a global health research firm, Saxenda, although it remained at the top in the local obesity treatment market, has seen its sales drop by 44.4 percent in the first quarter of this year to 5.9 billion won. In contrast, Qsymia rose to the position of the runner-up, recording 4.3 billion won in sales in the first three months of this year.
Saxenda’s first-quarter revenue marked a drop of more than 50 percent from its record sales of 11.9 billion won recorded in the third quarter of last year. If Qsymia continued to show strong sales growth, Saxenda would be at risk of losing its top place in the local obesity market since its launch in March 2018.
According to industry experts, there are various reasons for the success of Qsymia.
To start with, Qsymia showed a stronger weight control effect than conventional obesity treatments and improved convenience for patients as it is an oral treatment, which is considered to be plus for gaining popularity.
Another critical factor that has led to the success of Qsymia is the Korean regulators’ pulling Belviq, an obesity drug developed by Eisai and marketed by Ildong Pharmaceutical in Korea, off the shelves.
Belviq was the best seller in the local obesity treatment market in 2018, with a total sales of 9.8 billion won. Although the drug has since lost its top position to Novo Nordisk's Saxenda, it still had high demand among patients in Korea.
However, after the U.S. Food and Drug Administration warned of possible cancer risks and requested Eisai to take Belviq and Belviq XR off the market, the Ministry of Food and Drug Safety also recommended that Ildong Pharmaceutical recall it voluntarily.
Ildong has decided to accept the regulator's recommendation and withdraw the obesity drug.
Since then, drug companies staged a fierce competition to snatch the market share that had taken by Belviq as their own, and Alvogen's Qsymia seems to be a winner, at least so far, according to the watchers.
Except for Furimin, another obesity treatment of Alvogen Korea, most other obesity drugs performed poorly.
Sales for Daewoong Pharmaceutical's Dietamine, which was one of the top drugs in line to take Belviq's lost market shares, decreased 4.6 percent from the same period last year to 2.2 billion won, while those for Huon's Hutermin (1.4 billion won) and Alvogen Korea's Furing (1.2 billion won) also fell 0.2 and 3.8 percent, respectively.

