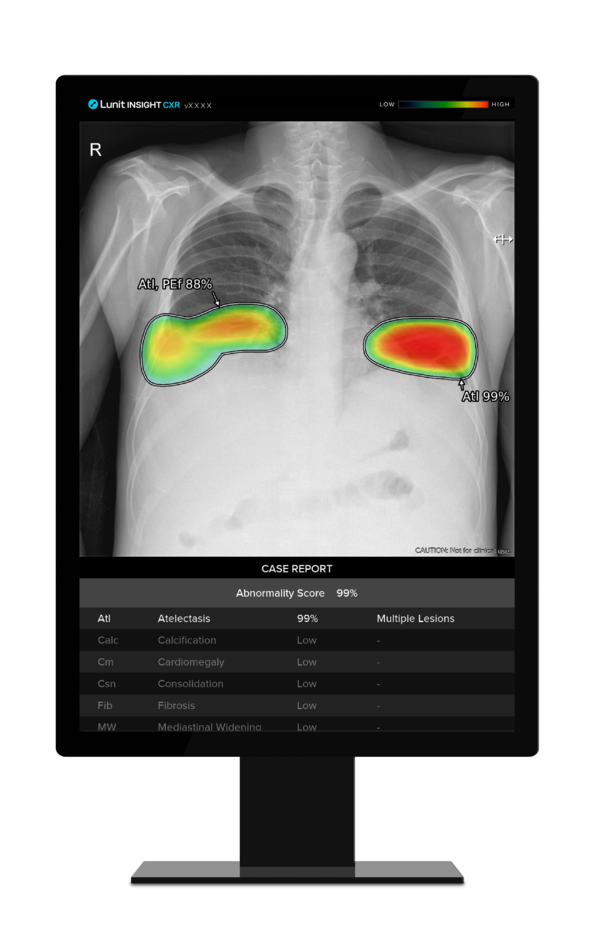
Lunit said its AI-powered chest screening solution, Lunit INSIGHT CXR, outperformed its competitors in a study comparing seven AI chest screening solutions.
The research was conducted by the Project AI for Radiology team at Radboud University Medical Center in the Netherlands, led by Professor Kicky G. van Leeuwen. The study analyzed thoracic X-ray data of 386 patients from seven Dutch hospitals, collected over a decade from Jan. 2012 to May 2022.
The team utilized seven different chest X-ray AI image analysis solutions in this study, pitting them against the interpretations of 13 radiologists and four radiology residents.
The seven solutions included Lunit and VUNO from Korea, Siemens Healthineers from Germany, Milvue from France, Oxipit from Lithuania, Annalise.ai from Australia, and Infervision from China.
The results showed that Lunit Insight CXR achieved an AUC (Area Under the Curve) score of 0.93, the highest among all solutions tested.
Generally, an AUC closer to 1 indicates superior performance, and above 0.8 is considered high-performing. The lowest AUC of 0.79 was noted for Infervision's InferRead DR Chest from China.
Lunit Insight CXR also demonstrated a higher sensitivity of 89 percent compared to both its AI counterparts and human radiologists, although its specificity of 80 percent was comparable to that of the human experts.
The company stressed that this balance of sensitivity and specificity is crucial in medical diagnostics, as it ensures both accurate detection and minimization of false positives and that the study focused on independently evaluating the ability of commercial AI solutions to detect pulmonary nodules and demonstrating the applicability and effectiveness of AI in a real-world medical setting.
Notably, the company explained that as the study found that four out of seven AI solutions exceeded the average reading ability of radiologists, AI will become an important tool in radiology in the near future.
"This study is significant as it represents the first third-party verification of the performance of Lunit Insight CXR," Lunit CEO Suh Beom-suk said. "Following a third-party verification paper published in JAMA Oncology in 2020, which acknowledged the superiority of Lunit Insight MMG over other competitors, Lunit has once again proven the world-class performance of all its products."
The results of the research were published in Radiology.
Related articles
- Lunit inks $1.86 million deal with Samsung to supply AI solutions for chest X-ray reading
- GC Cell, Lunit partner for CAR-NK cell therapy development with AI
- Lunit to acquire Volpara Health for $193 million to expand AI cancer screening in US
- Lunit publishes 'Lunit Scope IO' study in Nature’s sister publication
- Lunit secures 1st US sale of FDA-approved breast cancer detection solution
- Lunit's shares rebounding after record-breaking 2023 sales announcement
- Study on ‘Lunit SCOPE IO’ published in official SITC journal
- Lunit to supply AI solutions to medical institutions in Taiwan, Singapore
- VUNO registers US trademark for VUNO Med-DeepCARS

