Lunit, a healthcare AI company, said Tuesday that it has signed a contract with Samsung Electronics to supply AI solutions for chest X-ray reading.
The contract is valued at $1.86 million over three years, representing 17.7 percent of Lunit's 2022 sales.
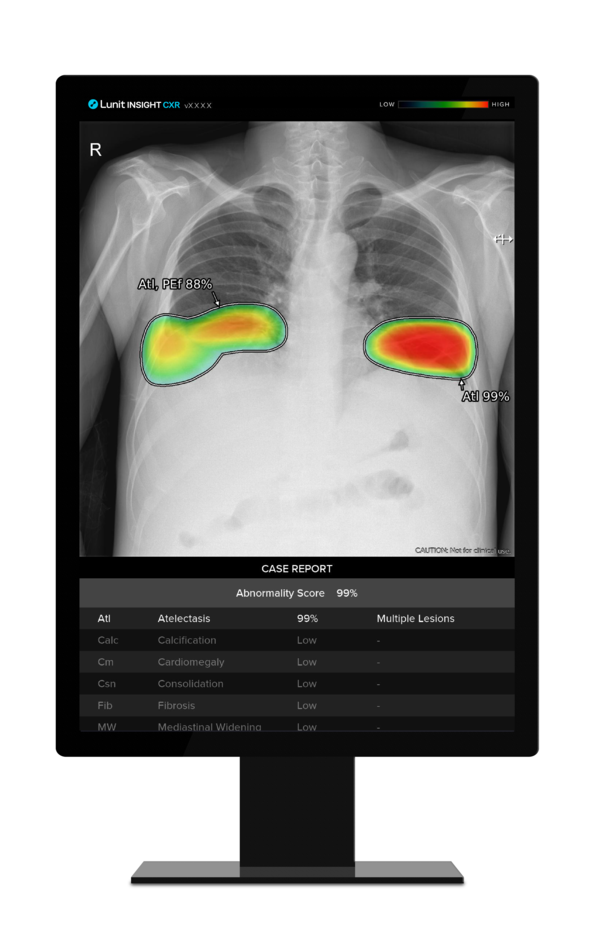
Under the agreement, Lunit will supply Lunit Insight CXR, an AI image analysis solution for chest x-rays, and Lunit Insight CXR Triage, an AI triage solution for emergency diseases, to Samsung Electronics, and Samsung Electronics will install Lunit's AI solutions on its premium x-ray machines and sell them in major countries.
Lunit Insight CXR is an AI solution that detects 10 chest diseases such as lung nodules, fibrosis, and calcification in chest X-ray images to assist medical professionals in diagnosing them.
Lunit Insight CXR Triage, an AI solution that automatically classifies normal and abnormal findings in emergency conditions such as pneumothorax and pleural fluid, was cleared by the FDA in November 2021.
The two companies plan to focus on selling X-ray devices equipped with AI solutions in North America, including the U.S., Canada, and Europe, while expanding their sales networks to the Middle East, South America, and Southeast Asia.
In particular, Lunit Insight CXR Triage will focus its sales efforts on hospital intensive care units and emergency departments, as it is strong in quickly and accurately classifying imaging information requiring emergency.
"The synergy between Lunit and Samsung Electronics will enable faster, more accurate chest screenings, leading to timely interventions and improved patient outcomes,” said Brandon Suh, CEO of Lunit. “We’re excited about the potential this partnership holds for advancing chest X-ray practices, particularly in ICUs and ERs."
Related articles
- Lunit publishes 'Lunit Scope IO' study in Nature’s sister publication
- Lunit's chest X-ray AI imaging solution to be reimbursable from March 2024
- Lunit to deploy AI chest screening to 5 Saudi hospitals, expanding presence in Middle East
- Lunit's AI-powered 3D breast tomosynthesis solution earns FDA clearance
- Lunit's AI-based lung nodule detection outperforms competitors
- Lunit secures 1st US sale of FDA-approved breast cancer detection solution
- Lunit's shares rebounding after record-breaking 2023 sales announcement
- Study on ‘Lunit SCOPE IO’ published in official SITC journal

