LifeSemantics said Thursday that it has completed the confirmatory clinical trial of its skin cancer image detection and diagnostic assistance software, a medical artificial intelligence solution that detects skin cancer.
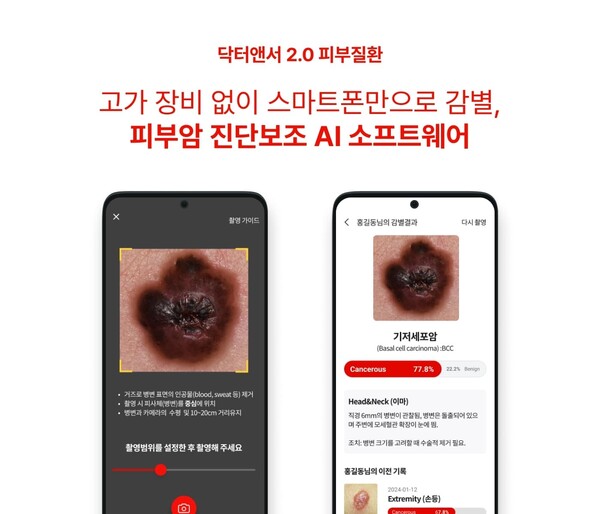
The clinical trial was a multi-center, retrospective study to prove the clinical superiority of the skin cancer diagnostic software medical device developed through the “Dr. Answer 2.0” project conducted by the National IP Industry Promotion Agency (NIPA).
Kyungpook National University Hospital, Keimyung University Dongsan Hospital, and Yeungnam University Medical Center collaborated. Researchers analyzed 199 cases in a confirmatory clinical trial of the algorithm, which was trained on 6,500 images of skin tumors of suspected skin cancer patients collected from each hospital.
LifeSemantics said the clinical trial showed that all metrics of sensitivity, specificity, and accuracy for differentiating between malignant and benign tumors exceeded minimum clinical performance standards, with a diagnostic accuracy rate of 80.9 percent.
The company expected the skin cancer diagnosis SW to help early detection and prevention of skin cancer by learning data on basal, squamous, and malignant melanoma. It also expected the new product to positively impact the adoption of medical AI and improve patient compliance by reducing the burden on primary medical institutions as it utilizes images taken with smartphones without expensive equipment.
"This is the first case in Korea to prove the performance of skin cancer AI through clinical trials," said Heo Eun-young, head of the R&D center at LifeSemantics. "We expect to increase access to treatment for skin cancer patients in Korea as we have confirmed that the results align with the results of clinical trials conducted overseas."
In addition to the skin cancer image detection and diagnosis aid software, LifeSemantics conducts clinical trials for home blood pressure prediction software, hypertension complication prediction software, and hair density analysis software.
The company also plans to finalize clinical trials and licensing to introduce the “canofyMD” lineup within the year.
Related articles
- LifeSemantics finishes technology demonstration of Dr.Call Thai
- LifeSemantics to reveal results for home blood pressure software trial in March
- ‘2024 will be 1st year of LifeSemantics’ entry into medical AI, DTx markets’
- LifeSemantics raises ₩20 billion ‘to step up overseas expansion’
- LifeSemantics gets nod for clinical trial of cardiovascular risk assessment AI

